Quantum Mechanics & Spectroscopy
Answers

Unit 57: Particle Waves
Q57.1) 0.24 nm
Q57.2) 6.63 x 10⁻²⁵ kg m/s
Q57.3) 0.7 nm
Q57.4) 3300 m/s
Q57.5) From the Rayleigh criterion, resolution is limited by diffraction. Smaller wavelengths diffract less, so result in better resolution (finer details). Optical microscopes use visible light with wavelengths of 400nm to 700nm, whereas electrons accelerated in microscopes have de Broglie wavelengths on the order of 0.01nm to 1nm. Since electrons have smaller wavelengths than visible photons, they diffract less and therefore result in better resolution.
Unit 58: Wavefunctions & Probability Density
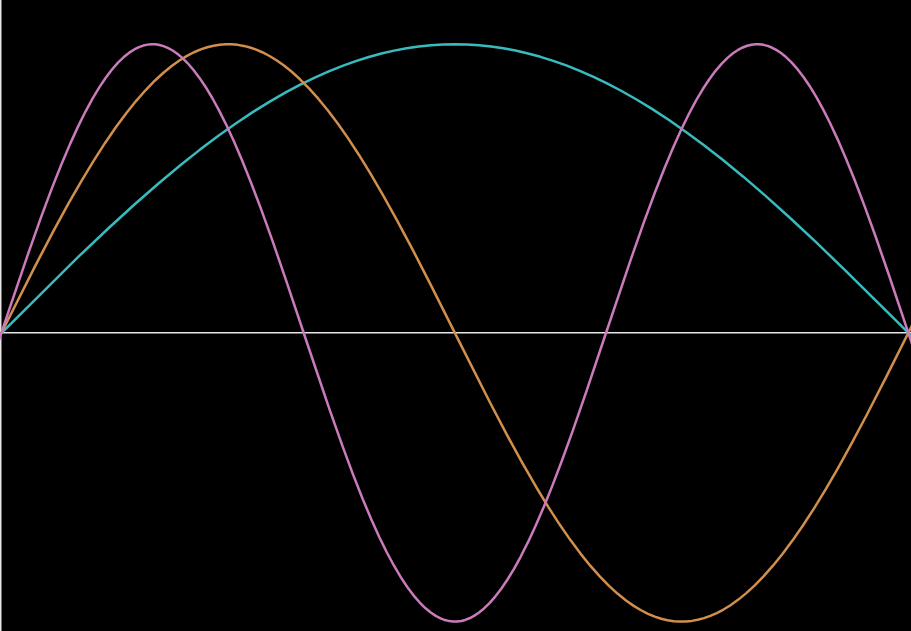
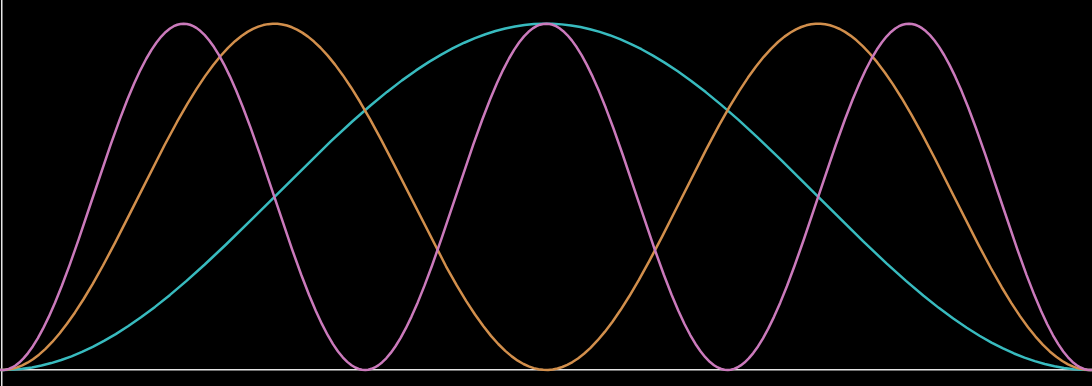
Q58.1)
a)
b)
c) x = L/6, L/2, 5L/6
d) x = 0, L/3, 2L/3, and L (ie near the 1st, 4th, 7th, and 10th carbons)
e) 0.9 nm
f) 1.48 nm⁻¹
g) 2.2%
h) 1/3 or 33.3%
Unit 59: π-electronic Energy Levels
Q59.1) a)
Ground state:
n=4 ─────────
n=3 ────↑↓────
n=2 ────↑↓────
n=1 ────↑↓────
1st excited state:
n=4 ─────↓───
n=3 ────↑────
n=2 ────↑↓────
n=1 ────↑↓────
b) n=1: 1.07×10⁻¹⁹ J, 0.67 eV
n=2: 4.28×10⁻¹⁹ J, 2.68 eV
n=3: 9.64×10⁻¹⁹ J, 6.02 eV
c) 4.28×10⁻¹⁹ J, 2.68 eV
Q59.2) a)
Ground state:
n = 2 ────── ──────
n = 1 ──↑↓─── ──↑↓───
n = 0 ──↑↓───
1st excited state:
n = 2 ───↓── ──────
n = 1 ──↑─── ──↑↓───
n = 0 ──↑↓───
b) n=1: 4.66×10⁻¹⁹ J, 2.91 eV
n=2: 1.86×10⁻¹⁸ J, 11.6 eV
n=3: 4.19×10⁻¹⁸ J, 26.2 eV
c) 1.86×10⁻¹⁸ J, 11.6 eV
Q59.3) a)
Ground state:
n=3 ────────
n=2 ────↑↓───
n=1 ────↑↓───
1st excited state:
n=3 ────↑────
n=2 ─────↓───
n=1 ────↑↓────
b) n=1: 2.68×10⁻¹⁸ J, 16.7 eV
n=2: 1.07×10⁻¹⁷ J, 66.9 eV
n=3: 2.41×10⁻¹⁷ J, 150 eV
c) 2.41×10⁻¹⁷ J, 150 eV
Q59.4) Since the energy of a level is proportional to n². the gaps between energy levels should increase as n increases, like this:
n=4 ────────
n=3 ────────
n=2 ────────
n=1 ────────
Q59.5) 0.75
Unit 60: Light Absorption by π-electrons in Biological Molecules
Q60.1)
1 → 2 transition = 3 eV
2 → 3 transition = 5 eV
1 → 3 transition = 8 eV
Q60.2)
1 → 2 transition = 413 nm
2 → 3 transition = 248 nm
1 → 3 transition = 155 nm
Q60.3) 133nm
Q60.4) 142nm
Q60.5) 407nm
Unit 61: Spectroscopy & the Beer-Lambert Law
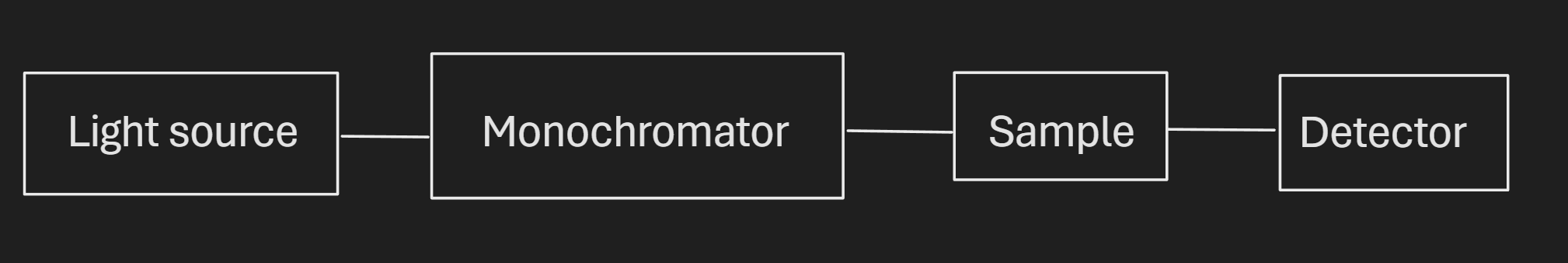
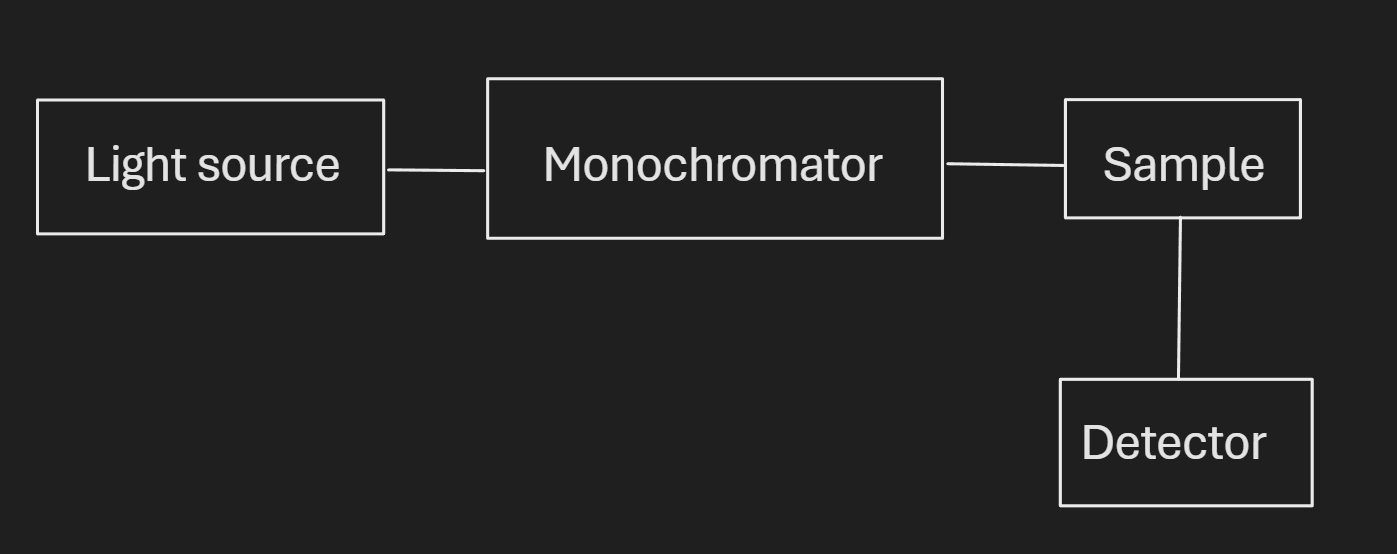
Q61.1)
Q61.2)
Q61.3) Both fluorescence and phosphorescence are types of photoluminescence (light emitted by a substance after it absorbs photons). The key difference is how the excited electrons return to the ground state and how long the emission lasts. Fluorescence is a very fast process; the excited electron stays in the excited state for nanoseconds. Phosphorescence is a comparatively slow process because the excited electron flips its spin, requiring an intersystem crossing before its able to relax.
Q61.4) a) 0.22
b) 0.6
c) 60%
Q61.5) 0.032%
Q61.6) 89.7m
Q61.7) a) 7
b) 3.5